Dear All (and with thanks to Lucy Naga for being the lead author on this economic-focused newsletter),
Antimicrobials are the quiet safety net of modern medicine. They underpin everything from routine infections to cancer treatment and surgery. They also support animal health and food production. Antimicrobial Effectiveness (AME, the ability of these medicines to remain efficacious and accessible for appropriate use) is a foundational component of the systems that sustain modern societies.
Despite this central role, AME is rarely treated as a strategic economic asset — and it was this economic perspective on antibiotics that was the starting point for the 6 Nov 2025 Panmure House Dialogues on Market Solutions for Antimicrobial Effectiveness. This dialogue took place at Panmure House, the final home of the moral philosopher and economist Adam Smith. Today, Panmure House serves as a center for economic and social research and debate, exploring contemporary challenges through the lens of Smith’s enduring ideas.
AMR is exactly the kind of problem Smith would have cared deeply about. While he championed the power of markets, he argued that they require well-designed institutions and aligned incentives to function in the public interest. For a thinker concerned with how nations flourish, the erosion of such a foundational resource would be an unmistakable call to action.
Against this backdrop, the dialogue brought together experts from healthcare, policy, academia, and finance to explore how economic actors—particularly investors—might help conserve and strengthen this vital resource.
— A shift in perspective —
What emerged from the discussion was a shift in perspective. AMR is usually framed as a health challenge. But AME also behaves as a form of infrastructure. It supports activity across multiple sectors, from healthcare to food systems, and its erosion creates risks that ripple through the wider economy.
Despite this, its value remains largely invisible in financial and corporate decision-making. The systems that shape investment, innovation and operational decisions rarely account for the benefits that effective antimicrobials provide or the risks associated with their loss.
The economic stakes are substantial. Research suggests that if antimicrobial resistance continues unchecked, global economic output could fall by around US$1.7 trillion annually by 2050 (27 Sep 2024 newsletter about McDonnell et al., 2024). At the same time, the economic case for action is strong: investments in conserving AME and improving access could generate returns many times greater than their cost (per other work by McDonnell et al., the 30-year payoff is 28:1. See the 29 Nov 2022 newsletter and its 8 Dec 2022 follow-up).
— Integrating this perspective into economic decision-making —
The challenge, then, is not awareness. The core challenge is integration: we must bring AME into the economic systems that shape real-world decisions.
To this end, the Panmure House Dialogue workshop has released a report entitled “Engaging Investors in Antimicrobial Effectiveness” that proposes a shared framework for action to help do exactly that. At its core is the idea that AME should be recognised explicitly as global economic infrastructure. If that framing is taken seriously, the need for 4 lines of work becomes evident:
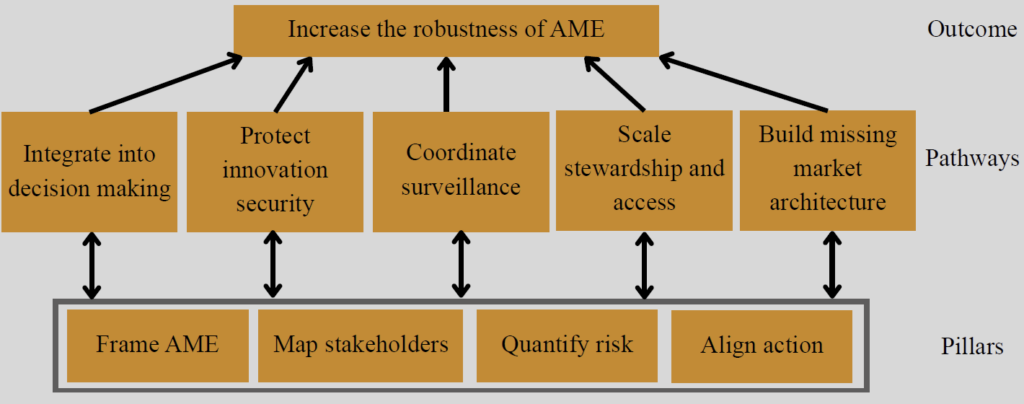
Shown at the bottom of the figure as Pillars, these actions are:
- Frame AME: AME should be framed as essential economic infrastructure to highlight its systemic value across sectors and scales.
- Map stakeholders: Actors who rely on or deplete AME need to be clearly mapped. These range from healthcare providers and pharmaceutical companies to agricultural producers, investors and regulators.
- Quantify risks: The operational, financial, and systemic risks faced by those actors as AME erodes should be estimated. Demonstrating the financial materiality of AME will lead relevant actors to include AME in their corporate strategies.
- Align action: All stakeholders that rely on and/or deplete AME should engage in established cross-sectoral dialogue to facilitate the sharing of information, alignment of incentives and coordination of targeted actions to conserve antimicrobial effectiveness.
— The role of markets —
With these insights, it is now clear why markets would enter the conversation. Markets shape how resources are valued, how capital is allocated, and how incentives are structured. If AME continues to sit outside those systems, it will remain undervalued and increasingly vulnerable. But if its role as infrastructure is recognised, markets could also become part of the solution.
The question is no longer simply how to fight AMR. It is how to build the institutional and economic architecture needed to protect one of the invisible foundations of modern life. Adam Smith’s Invisible Hand is a powerful tool in many parts of the marketplace, but it fails in the case of antibiotics. As Smith wrote in The Wealth of Nations (Book 5, Ch 1, Pt 3), “…the third and last duty of the sovereign…is that of erecting and maintaining those public institutions and those public works, which, though they may be in the highest degree advantageous to a great society, are, however, of such a nature that the profit could never repay the expense to any individual…”
— Making the invisible visible: Next steps —
As concluded in the report, “It is time to make the invisible visible. To realise these gains, AME-related risks and benefits must be identified, their financial materiality quantified, and their role embedded within investment, corporate and policy decision-making. This requires a granular understanding of how value is created, preserved and eroded across interconnected systems, combined with clear pathways through which different actors can contribute to conserving AME.”
Excitingly, the 6 Nov 2025 workshop is only the first of a Panmure House series of discussions of this topic. The focus of these workshops will be on the actions needed to build the institutional and economic architecture needed to protect one of the invisible foundations of modern life. And if you want to do a bit more reading on the economics of antibiotics, a short list of additional resources is found below our signatures.
All best wishes, Lucy (with a bit of help from John and as well a shout-out to Deepali Patel, Director, International Policy at the AMR Action Fund for critical commentary)
Lucy Naga, PhD | Research Associate in Economics | Heriot-Watt University | Manager, Panmure House Dialogues on Market Solutions for Antimicrobial Effectiveness | L.Naga@hw.ac.uk
John H. Rex, MD | Chief Medical Officer, F2G Ltd. | Operating Partner, Advent Life Sciences. Follow me on Twitter: @JohnRex_NewAbx. See past newsletters and subscribe for the future: https://amr.solutions/blog/. All opinions are my own.
For further reading on the ideas behind antimicrobial effectiveness, please see:
- Papers focused on economics
- 19 Mar 2024 newsletter entitled “Tragedy of the (Antibiotic) Commons: A true market failure”
- 28 Feb 2024 newsletter entitled “How economists (and Finance Ministers) think…”
- Brown G and Layton DF (1996): ‘Resistance Economics: social cost and evolution of antimicrobial resistance’. Environ Develop Econ. 1(3):349-355. doi:10.1017/S1355770X0000067X. This was one of the first papers to formalize the economics of AMR, highlighting market failure.
- Weldon I, Liddell K, et al. (2024): ‘Analyzing Antimicrobial Resistance as a series of Collective Action Problems’. Policy Studies J 52(4):833-856. https://doi.org/10.1111/psj.12552. This paper’s approach to a game-theoretic understanding gives an interesting insight into the dynamics of AMR and the complexity of systems.
- Reports on the role of investors in AMR
- The Shareholder Commons (2022): ‘Antimicrobial Resistance and the Engagement Gap: why investors must do more to move the needle.’ This whitepaper considers the ways that AMR affects the long-term returns of diversified portfolios, which are affected by systemic risks, and suggests investor-determined guardrails on AMU (which would require collective action).
- IAAMR (2022): ‘Investor Action on AMR Progress Report.’ This whitepaper includes case studies of what investors are doing to account for AMR.
- FAIRR (2024): ‘Health and Wealth: the Investors’ Guide to AMR, a growing health crisis.’ This whitepaper identifies AMR as systemic risk, outlines economic case for investment in AMR solutions.
- Discussions of the problem of access
- 19 Aug 2021 newsletter entitled “Ex-US new antibiotic access: Multi-year delays, even in Europe!”
- 1 Dec 2024 newsletter entitled “The 6 meanings of ‘Lack of Access’ (UNSLAP)”
- 7 Apr 2025 newsletter entitled “UNSLAP: You reach for the antibiotic … and it’s not there!”
John’s Top Recurring Meetings
Virtual meetings are easy to attend, but regular attendance at annual in-person events is the key to building your network and gaining deeper insight. My personal favorites for such in-person meetings are below. Of particular value for developers, the small meeting format of BEAM’s AMR Conference (March) and GAMRIC (September-October; formerly, the ESCMID-ASM conference series) creates excellent global networking. IDWeek (October) and ECCMID (April) are much larger meetings but also provide opportunities for networking with a substantial, focused audience via their Pipeline sessions. Hope to see you there!
- 17-21 April 2026 (Munich, Germany): ESCMID Global 2026, the annual meeting of the European Society for Clinical Microbiology and Infectious Diseases. You can go here to register and view the preliminary program; the abstract submission window for 2026 will run 15 October to 26 Nov 2025. For those who would like a substantial opportunity to present a product to a large audience (see also adjacent note about IDWeek), I know that the meeting schedule will again include Pipeline Monday.
- [ABSTRACT DEADLINE is Fri 10 Apr at noon CET] 22-24 Sep 2026 (Lisbon, Portugal): The 2nd GAMRIC, the Global AMR Innovators Conference (London, UK). Formerly the ESCMID-ASM (or ASM-ESCMID depending on location) Joint Conference on Drug Development for AMR, 2026 will be the 11th year for this series that is now under the joint sponsorship of CARB-X, ESCMID, BEAM Alliance, GARDP, LifeArc, Boston University, and AMR.Solutions. The ongoing series employs the successful format of prior meetings with a single-track meeting and substantial networking time. The 2025 meeting was a sell-out success! A written summary of the 2025 meeting is here and the videos from the sessions are now available here. Registration for the 2026 meeting is now open and abstract submissions are open through Fri 10 Apr at noon CET. Note as well that a late-breaker window be announced for July.
- 21-24 Oct 2026 (Washington, DC, USA): IDWeek 2026, the annual meeting of the Infectious Diseases Society of America. Details are not yet available but I would expect the program to continue to provide a substantial opportunity to present a product to a large audience (see also adjacent note about ESCMID) as well as opportunities to present at an IDWeek Pipeline Session.
- 23-24 Mar 2027 (Basel, Switzerland): The 10th AMR Conference (3-4 Mar 2026) is now over and offered a rich program that included a 10-year retrospective (we’ve done a lot!), regulatory updates, discussions of how to pursue development in China, and much more … in addition to being a superb opportunity for networking! I am told the session videos will soon be available on the conference website. Mark your calendars for next year’s 11th AMR Conference!
Upcoming meetings of interest to the AMR community:
- 24 Mar 2026 (1-2p ET, virtual): CDC webinar entitled “Tools to Tell the Story: New Resources for Communicating about Antimicrobial Resistance.” Go here for additional details to register,
- 26 Mar 2026 (12.00-13.30 ET, virtual): REVIVE (GARDP)-sponsored webinar: “Journal Club: Key findings from recent publications in antimicrobial R&D.” Go here for details and to register.
- 17-21 April 2026 (Munich, Germany): ESCMID Global 2026, the annual meeting of the European Society for Clinical Microbiology and Infectious Diseases. See Recurring Meetings list, above.
- 4-8 June 2026 (Washington, DC): ASM Microbe, the annual meeting of the American Society for Microbiology. The meeting format is evolving and next year will combine 3 meetings (ASM Health, ASM Applied and Environmental Microbiology, and ASM Mechanism Discovery) into one event. Go here for details.
- 11-12 Jun 2026 (Washington, DC): The Second Annual Unite for Sepsis Symposium, presented by the Sepsis Alliance. The event seeks to accelerate progress in sepsis research and care. Go here for details and to register.
- 22-24 Sep 2026 GAMRIC (Lisbon, Portugal), the Global AMR Innovators Conference (London, UK; formerly the ESCMID-ASM Joint Conference on Drug Development for AMR). See list of Top Recurring meetings, above..
- [REGISTRATION NOW OPEN] 10-18 Oct 2026 (Annecy, France, residential in-person program): ICARe (Interdisciplinary Course on Antibiotics and Resistance) … and 2026 will be the 10th year for this program. Patrice Courvalin orchestrates content with the support of an all-star scientific committee and faculty. The resulting soup-to-nuts training covers all aspects of antimicrobials, is very intense, and routinely gets rave reviews! Registration for 2026 will be during March 16–July 3, 2026. You can go here for more details and you should put a reminder in your calendar register on / after 16 March.
- 21-24 Oct 2026 (Washington, DC, USA): IDWeek 2026. See list of Top Recurring meetings, above.
- 10-13 November 2026 (Madrid, Spain): The International Society for Infectious Diseases (ISID) has announced its 21st International Congress on Infectious Diseases (ICID). Register and view the preliminary program here (Early bird closes 30 July 2026); abstract deadline is 28 April 2026.
- 23-24 Mar 2027 (Basel, Switzerland): The 11th AMR Conference sponsored by the BEAM Alliance. See list of Top Recurring meetings, above.
- ??? Mar 2028 (yes, that’s 2028, with location TBD): The 2028 Gordon Research Conference (GRC, https://www.grc.org/) entitled “Antibacterials of Tomorrow to Combat the Global Threat of Antimicrobial Resistance” and its related Gordon Research Seminar (GRS) for young doctoral and post-doctoral researchers will be sometime in March 2028. The organizers hope to coordinate dates and location with the 2028 BEAM-AMR meeting. Details to follow — mark your calendar!
Self-paced courses, online training materials, and other reference materials:
- OpenWHO: “Antimicrobial Resistance in the environment: key concepts and interventions.” Per the webpage for the course, it will teach you “…why addressing AMR in the environment is essential and gain insights into how action can be taken to prevent and control AMR in the environment at the national level.” This course builds on WHO’s 2024 Guidance on wastewater and solid waste management for manufacturing of antibiotics. For further reading, see also the 25 Sep 2023 newsletter entitled “Manufacturing underpins both access and stewardship: Cefiderocol as a case study” and the 28 Jan 2024 newsletter entitled “EMA Concept Paper: Guidance on manufacturing of phage products”.
- GARDP’s REVIVE website provides an encyclopedia covering a range of R&D terms, recordings of prior GARDP webinars, a variety of viewpoint articles, and more! Check it out!
- GARDP’s https://antibioticdb.com/ is an open-access database of antibacterial agents.
- The CARB-X website provides a range of recordings from its webinars, bootcamps, and more. A bit of browsing would be time well spent!
- British Society for Antimicrobial Chemotherapy offers an eLearning section: Education – The British Society for Antimicrobial Chemotherapy.
- CARB-X have announced a funding round that will be open 8-22 April 2026 (there will also be a round during 4Q 2026). There are 4 funding themes as below. Informational webinars have been announced for 9 and 10 April 2026.
- Direct-acting therapeutics for infections caused by Gram-negative bacteria
- Novel Chemistry for AMR Challenge – target-based therapeutics.
- Non-vaccine approaches to prevent neonatal sepsis
- Diagnostics for neonatal sepsis
- The Horizon Europe Work Programme 2026-2027 includes at least 3 calls of interest within its Cluster 1 — see the list below. The application window starts 10 Feb 2026 and closes on 16 Apr 2026. See also the 12 Dec 2025 newsletter about the call. Note as well that there calls for agents to prevent and/or treat viral infections.
- HORIZON-HLTH-2027-01-DISEASE-08: Development of innovative antimicrobials against pathogens resistant to antimicrobials
- HORIZON-HLTH-2027-02-IND-02: Portable point-of-care diagnostics
- HORIZON-HLTH-2026-01-DISEASE-03:Advancing research on the prevention, diagnosis, and management of post-infection long-term conditions.
- ENABLE-2 has continuously open calls for both its Hit-to-Lead program as well as its Hit Identification/Validation incubator. Applicants must be academics and non-profits in Europe due to restrictions from the funders. Applications are evaluated in cycles … see the website for details on current timing for reviews.
- BARDA’s long-running BAA (Broad Agency Announcement) for medical countermeasures (MCMs) for chemical, biological, radiological, and nuclear (CBRN) threats, pandemic influenza, and emerging infectious diseases is now BAA-23-100-SOL-00004 and offers support for both antibacterial and antifungal agents (as well as antivirals, antitoxins, diagnostics, and more). Note especially these Areas of Interest: Area 3.1 (MDR Bacteria and Biothreat Pathogens), Area 3.2 (MDR Fungal Infections), and Area 7.2 (Antibiotic Resistance Diagnostics for Priority Bacterial Pathogens). Although prior BAAs used a rolling cycle of 4 deadlines/year, the updated BAA released 26 Sep 2023 has a 5-year application period that ends 25 Sep 2028 and is open to applicants regardless of location: BARDA seeks the best science from anywhere in the world! See also this newsletter for further comments on the BAA and its areas of interest.
- HERA Invest was launched August 2023 with €100 million to support innovative EU-based SMEs in the early and late phases of clinical trials. Part of the InvestEU program supporting sustainable investment, innovation, and job creation in Europe, HERA Invest is open for application to companies developing medical countermeasures that address one of the following cross-border health threats: (i) Pathogens with pandemic or epidemic potential, (ii) Chemical, biological, radiological and nuclear (CBRN) threats originating from accidental or deliberate release, and (iii) Antimicrobial resistance (AMR). Non-dilutive venture loans covering up to 50% of investment costs are available. A closing date is not posted insofar as I can see — applications are accepted on a rolling basis; go here for more details.
- The AMR Action Fund is open on an ongoing basis to proposals for funding of Phase 2 / Phase 3 antibacterial therapeutics. Per its charter, the fund prioritizes investment in treatments that address a pathogen prioritized by the WHO, the CDC and/or other public health entities that: (i) are novel (e.g., absence of known cross-resistance, novel targets, new chemical classes, or new mechanisms of action); and/or (ii) have significant differentiated clinical utility (e.g., differentiated innovation that provides clinical value versus standard of care to prescribers and patients, such as safety/tolerability, oral formulation, different spectrum of activity); and (iii) reduce patient mortality. It is also expected that such agents would have the potential to strongly address the likely requirements for delinked Pull incentives such as the UK (NHS England) subscription pilot and the PASTEUR Act in the US. Submit queries to contact@amractionfund.com.
- INCATE (Incubator for Antibacterial Therapies in Europe) is an early-stage funding vehicle supporting innovation vs. drug-resistant bacterial infections. The fund provides advice, community, and non-dilutive funding (€10k in Stage I and up to €250k in Stage II) to support early-stage ventures in creating the evidence and building the team needed to get next-level funding. Details and contacts on their website (https://www.incate.net/).
- These things aren’t sources of funds but would help you develop funding applications
- The Global AMR R&D Hub’s dynamic dashboard (link) summarizes the global clinical development pipeline, incentives for AMR R&D, and investors/investments in AMR R&D. See also the 7 Feb 2026 newsletter (“The global funding pipeline, 2017-2023: A review”) about an excellent deep dive by the Hub team into patterns of funding over time.
- Antimicrobial Resistance Research and Innovation in Australia is an actively updated summary that covers Australia’s AMR research and patent landscape. It is provided via collaboration between The Lens (an ambitious project seeking to discover, analyse, and map global innovation knowledge) and CSIRO (Commonwealth Scientific and Industrial Research Organisation, an Australian Government agency responsible for scientific research). Lots to explore here!
- Diagnostic developers would find valuable guidance in this 6-part series on in vitro diagnostic (IVD) development. Sponsored by CARB-X, C-CAMP, and FIND, it pulls together real-life insights into a succinct set of tutorials.
- In addition to the lists provided by the Global AMR R&D Hub, you might also be interested in my most current lists of R&D incentives (link) and priority pathogens (link).