Dear All,
Continuing on the theme of helping countries develop their own national action plans, WHO released the “WHO implementation handbook for national action plans on antimicrobial resistance: guidance for the human health sector“. The handbook has some great infographics like the one below and will shortly be translated into French, Spanish, and Russian.
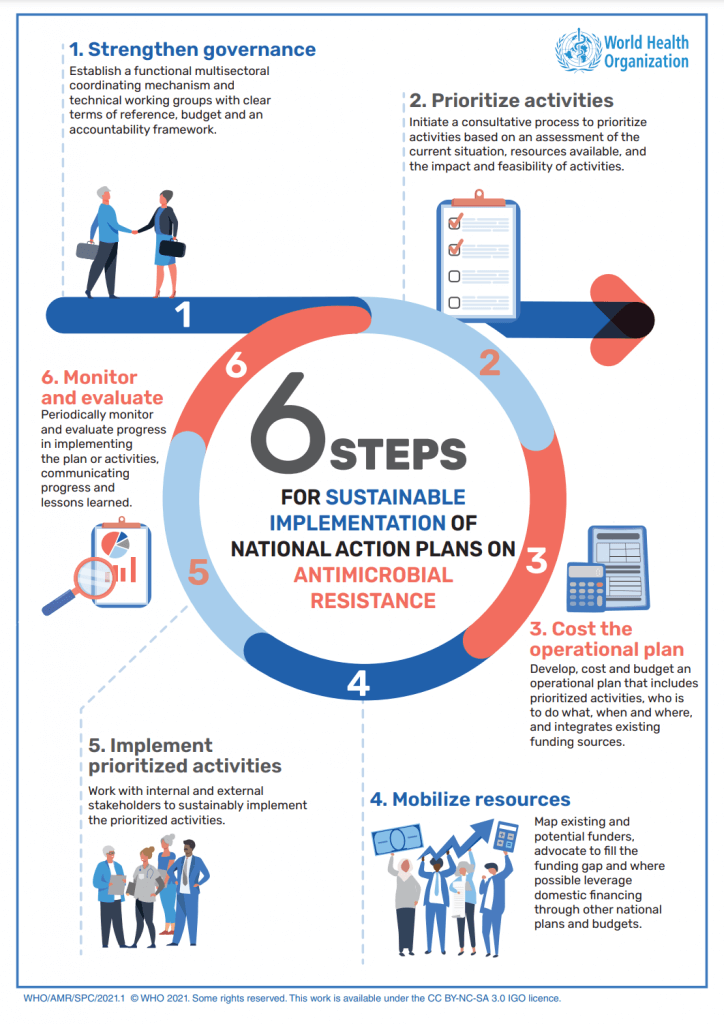
There will be an official launch on Thursday 17 March 2022 with two webinars at 11a CET and 4p CET respectively. The webinars have the same basic content but different time zones. Registration is now open and the current schedule is as follows:
- Opening remarks: WHO HQ and WHO regional offices
- WHO Implementation handbook for national action plans on AMR: Dr Sarah Paulin-Deschenaux, Technical officer, AMR division, WHO HQ
- Regional and country experiences:
- Session 1: Regional experience and lessons learned from Africa on national action plan implementation: Dr Laetitia Gahimbare, Regional AMR focal point, WHO AFRO
- Session 2: Regional experience and lessons learned from the Americas on national action plan implementation: Dr Pillar Ramon-Pardo, Regional AMR advisor, PAHO/WHO AMRO
- Country experiences:
- Session 1: Country experience on challenges, enablers and lessons learned with national action plan on AMR implementation: Mali and Tanzania
- Session 2: Country experience on challenges, enablers and lessons learned with national action plan on AMR implementation: TBC
- A moderated Q&A will follow the presentations
- Moderator for session 1: Dr Otrida Kapona, WHO AMR STAG member, Zambia
- Moderator for session 2:TBC
As I hope you know, National Action Plans are being steadily created (as of Oct 2021, WHO reported that 48 countries had finalized their NAP). You can also track NAP activity via the Global Database for the Tripartite Antimicrobial Resistance (AMR) Country Self-assessment Survey (TrACSS).
But, creating the plan is the (relatively) easy part … the hard work is in making it happen. To that end, you should be interested in this handbook (and the webinars) if you are a national or subnational stakeholders working on AMR within the human health sector. This includes “national health authorities, national multisectoral coordination groups, senior technical experts and policymakers involved in implementing AMR activities at all levels of the health system, and implementation partners to accelerate sustainable implementation and M&E of NAPs on AMR.”
Good work, Team WHO! All best wishes, –jr
John H. Rex, MD | Chief Medical Officer, F2G Ltd. | Operating Partner, Advent Life Sciences. Follow me on Twitter: @JohnRex_NewAbx. See past newsletters and subscribe for the future: https://13.43.35.2/blog/. All opinions are my own.
Current funding opportunities (most current list is here):
- [NEW] ICARS– and IDRC-sponsored RFP on gender inequality in AMR (with a focus on sub-Saharan Africa) for proposals of up to $150k for a 9-12 month project. Deadline for proposals is 9 Mar 2022; go here for details.
- NIAID has released a four-pronged BAA (Broad Agency Announcement (HHS-NIH-NIAID-BAA2022-1) that coverstherapeutics (antibacterial, antifungal, antiviral), vaccines (again, all 3 areas), and diagnostics. Lots of possibilities! The due date is 18 Mar 2022.
- BARDA is seeking proposals under Project BioShield to support late stage development, regulatory approval and potential procurement of antibiotics against biothreat pathogens, Yersinia pestis, Francisella tularensis, and Burkholderia pseudomallei. Small businesses can apply until April 6, 2022. Go here for the details.
- JPIAMR’s 14th call is now open. Entitled “Disrupting drug resistance using innovative design”, the call seeks consortia that would seek to “improve the treatment of bacterial and fungal infections (including co-infection) and/or the prevention of the emergence/spread of resistance in humans, animals or plants through the improvement of the efficacy, specificity, delivery, combinations and/or repurposing of drugs and plant protection agents.” Bacteria, fungi, human health, animal health, and plant health are all in scope! Pre-proposals are due 8 Mar 2022; full proposals would be due 5 July 2022. Go here for details.
- The AMR Action Fund is now open to proposals for funding of Phase 2 / Phase 3 antibacterial therapeutics. Per its charter, the fund prioritizes investment in treatments that address a pathogen prioritized by the WHO, the CDC and/or other public health entities that: (i) are novel (e.g., absence of known cross-resistance, novel targets, new chemical classes, or new mechanisms of action); and/or (ii) have significant differentiated clinical utility (e.g., differentiated innovation that provides clinical value versus standard of care to prescribers and patients, such as safety/tolerability, oral formulation, different spectrum of activity); and (iii) reduce patient mortality. It is also expected that such agents would have the potential to strongly address the likely requirements for delinked Pull incentives such as the UK (NHS England) subscription pilot and the PASTEUR Act in the US. Submit queries to contact@amractionfund.com.
- INCATE (Incubator for Antibacterial Therapies in Europe) is a newly launched early-stage funding vehicle. Details are still coming into focus, but per comments on 25 Aug 2021 at the BIOCOM conference, their goal is to support ~4 companies per year with about $250k/company. Contact details are on their website (https://www.incate.net/).
- CARB-X recently announced that their existing resources will be reserved to fund their existing portfolio (more than 80 total awards, and counting, as they include contracting from prior rounds). New rounds from CARB-X will occur only after new funding is obtained in 2021.
- It’s not a funder, but AiCuris’ AiCubator offers incubator support to very early stage projects. Read more about it here.
- The Global AMR R&D Hub’s dynamic dashboard (link) summarizes the global clinical development pipeline, incentives for AMR R&D, and investors/investments in AMR R&D.
- In addition to the lists provided by the Global AMR R&D Hub, you might also be interested in my most current lists of R&D incentives (link) and priority pathogens (link).
Upcoming meetings of interest to the AMR community (most current list is here):
- [If you missed it, you can now watch the video] 8 Dec 2021: “The New Winds Pushing and Pulling Antibacterial Development.” This FABULOUS program featured talks from the UK team behind the NHS “Netflix” pilot, Kevin Outterson’s recently released report documenting the need for global Pull incentives to have a value of $2.2 – 4.8b, and speakers covering PASTEUR and work in the EU on pull incentives. The video is here — please make time to listen to this program!
- [GRAM report launch video!]: The 4 Feb 2022 webinar for the GRAM (Global Research on Antimicrobial Resistance) report (“1.27 million deaths per year are directly attributable to AMR”) is now available for replay. #AMRSOS!
- 23 Feb 2022 (virtual, 8-9a EST): Pre-application webinar to provide more information and to answer questions. Register here.
- 3 Mar 2022 (virtual, 2p GMT): The Longitude Prize (run by Nesta Challenges), CARB-X and FIND, have joined forces for a virtual event in March, AMR& Diagnostic Frontiers: Developing and increasing access to innovative diagnostics in and for low-and-middle-income countries (LMICs). Full agenda details and registration can be found here, including an invitation for diagnostic companies to pitch to a panel of global health representatives.
- 3-6 Mar 2022 (Albuquerque, New Mexico): Biannual meeting of the MSGERC (Mycoses Study Group Education and Research Consortium). Details are here.
- 6-11 Mar 2022 (Il Ciocco, Tuscany): Gordon Research Conference entitled “New Antibacterial Discovery and Development”. Go here for details, go here for the linked 5-6 Mar Gordon Research Seminar that precedes it.
- 9 Mar 2022 (virtual, and in-person): BioInfect Conference, Alderley Park, UK (near Manchester). This long-running Bionow-sponsored annual conference draws a very strong audience. Go here for details.
- [NEW] 23-24 Mar 2022 (virtual): JPIAMR workshop entitled “Supporting research and innovation on One Health AMR Surveillance”. Go here for details.
- 7-8 Apr 2022 (Basel and in person, we hope): The 6th edition of the annual AMR conference sponsored by the BEAM Alliance, CARB-X, the Novo REPAIR Impact Fund, the IMI Accelerator, and the European Biotechnology Network. Go here for the hold-the-date page and a way to be kept informed about the meeting.
- 9-13 May 2022 (Athens and online): 40th Annual Meeting of the European Society for Paediatric Infectious Diseases, Go here for details.
- 20-24 Sep 2022 (New Delhi): 21st Congress of the International Society for Human and Animal Mycology (ISHAM). Go here for details.
- 25-28 Oct 2022 (Stellenbosch, South Africa): The University of Cape Town’s H3D Research Centre will celebrate its 10th anniversary with a symposium covering the Centre’s research on Malaria, TB, Neglected Tropical Diseases, and AMR. Go here to register.