Dear All (and with thanks to Aaron Dane for co-authoring this newsletter AND inhale, exhale … this is going to be a long, wonkish, but VERY important note! Get coffee! Get to a calm place! Clear your head! Read on!),
Provoked by a recent paper on the idea of DTR (Difficult-to-Treat Resistance) by Walker et al. (from the group led by Sameer Kadri at the NIH Clinical Center, full details on the paper are below), we are today going to visit the intersection of a set of intertwined themes. Deep, deep down, this conversation is about how we know whether (or not) new drugs really work for bad bugs. Yes, we can see the favorable microbiology in the laboratory. Yes, we can see the favorable results of animal models. But, what about in human infections? “I want to see data on new drugs for bad bugs in people,” you cry! “Why are you bringing me these confusing non-inferiority results? Why aren’t you studying bad bugs? And, superiority studies would be cleaner! They can be smaller! Why aren’t we instead seeing superiority data on treatment of bad infections?”
Well, there are good explanations for (i) why we can know that new antibiotics will work on bad bugs and (ii) why superiority studies are NOT routine. These ideas have been covered in prior newsletters, but it seems a good time to draw all these threads into an updated, comprehensive summary.
Our tour today is going to have 7 stops:
- (Context) How do we classify resistant bacteria? I’ve heard of MDR and XDR, but what are UDR, PDR, and DTR?
- (Use of new agents) Since the idea of DTR emerged in the mid-20-teens, we have at least some new, safe beta-lactam antibiotics. Are these newer agents being used for DTR infections?
- (Impact of new agents: the new paper is discussed here!) What is the impact of these newer agents on outcome in DTR infections?
- (One step further) How much of a mortality improvement is actually plausible?
- (Implications, Part 1) Can we ever show that new drugs are better?
- (Implications, Part 2) What does this mean for calls to improve outcomes through alternative approaches such as host-directed therapies?
- (Implications, Part 3) Rapid diagnostics and access to DTR-active agents are the keys to taking action!
Know that as we write we will sometimes be repeating materials from prior newsletters … you can see the full list of sources below our signatures. With that, here we go!
——————————
Stop #1 (Context): How do we classify resistant bacteria? I’ve heard of MDR and XDR, but what are UDR, PDR, and DTR?
It’s helpful to see the spectrum of resistance as having 5 categories:
- UDR (Usual Drug Resistance): Any resistance profile that is not MDR, XDR, or PDR. This is the current expected pattern of susceptibility to available agents. There will be non-susceptibility to some agents but first-line therapies are reliably active.
- MDR (Multi-Drug Resistance): non-susceptibility to at least one agent in three or more antimicrobial categories.
- XDR (eXtensive Drug Resistance): non-susceptibility to at least one agent in all but two or fewer antimicrobial categories (i.e., bacterial isolates remain susceptible to only one or two categories).
- PDR (Pan Drug Resistance): non-susceptibility to all agents in all antimicrobial categories.
- DTR (Difficult-to-Treat Resistance): non-susceptibility to all of the typical first-line, lower toxicity agents.
- Sources:
- The definition of UDR is an idea that a group (including both Aaron and me) coined and discussed in McDonnell et al. CID 63:(Suppl. 2): S57–9, 2016 and Rex et al. CID 65:141-6, 2017).
- CDC and ECDC gave us the ideas of MDR, XDR, and PDR in Magiorakos et al. CMI 18:268-81, 2012).
- Finally, the idea of DTR was proposed in Kadri et al. CID 67:1803-14, 2018.
The first 4 categories (UDR to PDR) are step-wise variants of each other. UDR is, well, the usual thing — what you expect. The organism may have some resistance mechanisms but standard first-line therapies are reliable. Therapeutic choices are narrowed as you go from MDR (choose carefully!) to XDR (only 1 or 2 choices) to PDR (no choices).
The concept of DTR is, however, different from the stepwise ladder of UDR-MDR-XDR-PDR. The idea is that the UDR-MDR-XDR-PDR definitions make no distinction between strengths and weaknesses of the individual antibiotics: agents with higher efficacy and lower toxicity are counted in the same way as agents with lower efficacy and higher toxicity. So, DTR is defined as resistance to all of the typical first-line, lower toxicity agents. The organism might really be just MDR if you just count active drugs, but Kadri et al. argue resistance to fluoroquinolones and the beta-lactams (including carbapenems and beta-lactamase inhibitor combinations) is DTR in that you are forced to use less desirable agents with either toxicity (e.g., the nephrotoxic aminoglycosides) or other pharmacology limitations.
If you’d like a bit more detail on this key idea, there’s a 5-minute YouTube explainer of the concept as well as this diagram:
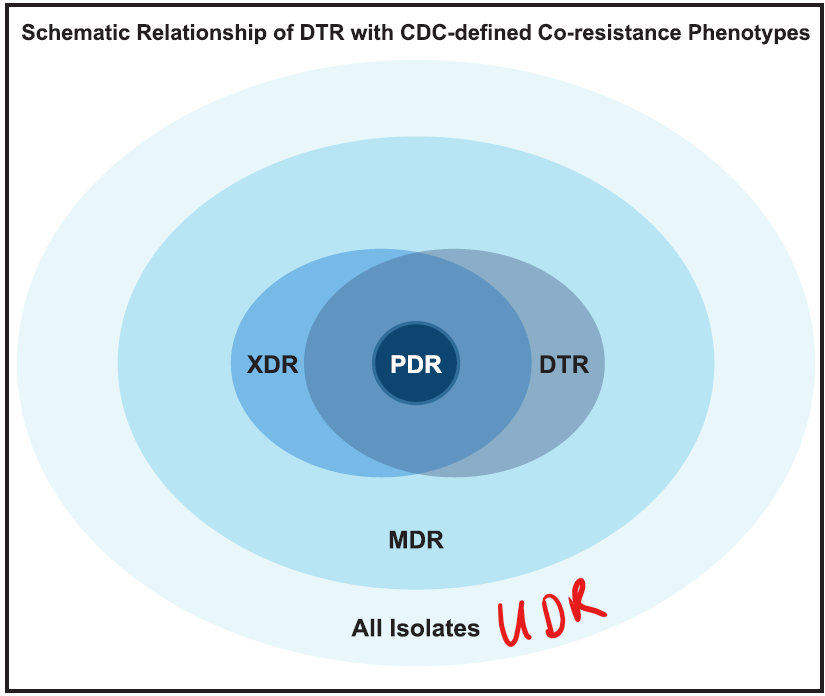
Diagram of the concept of DTR from Kadri 2018. Note that DTR includes only portions of MDR or XDR.
The idea of discriminating among the qualities of the agents was shown to be important by Kadri et al. 2018. Based on data from the Premier Healthcare Database from 173 US institutions on 46,521 Gram-Negative Blood Stream Infection (GNBSI) isolates from the period 2009-2013, about 1.5% of isolates were DTR. Critically, patients with DTR GNBSI isolates had a 40% higher adjusted mortality risk than those with non-DTR GNBSI. These findings were consistent in a variety of sensitivity analyses including by-species analyses and were also further validated using detailed clinical data in Kadri et al. OFID 6:ofz110, 2019.
——————————
Stop #2 (Use of new agents): Since the idea of DTR emerged in the mid-20-teens, we have at least some new, safe beta-lactam antibiotics. Are these newer agents being used for DTR infections?
OK, so DTR is associated with increased mortality! But when you have an active beta-lactam, things should improve. The DTR data above were for the period 2009-2013. Since then, we’ve had new agents, especially new beta-lactams reach approval. Are these agents being used?
Well, sometimes. For data, we need to consider 2 papers and a related editorial:
- Outterson K et al. (2021): Patient access in fourteen high-income countries to new antibacterials approved by the FDA, EMA, PMDA, or Health Canada, 2010-2020. Clinical Infectious Diseases. 2021;74:1183-90. https://doi.org/10.1093/cid/ciab612.
- Strich et al. (2024): Assessing Clinician Utilization of Next-Generation Antibiotics Against Resistant Gram-Negative Infections in U.S. Hospitals: A Retrospective Cohort Study. Ann Intern Med. 2024;177:559-72, https://doi.org/10.7326/M23-23
- And the excellent associated commentary on Strich 2024 by Howard-Anderson and Boucher (2024): New Antibiotics for Resistant Infections: What Happens After Approval? Annals of Internal Medicine. 2024:177:674-5, https://doi.org/10.7326/M24-0192).
The first paper (Outterson 2021) has an associated newsletter entitled “Ex-US new antibiotic access: Multi-year delays, even in Europe!”. In brief, this paper found that access to newer antibiotics was often delayed for years, even in high-income countries. Only 3 countries (United States, United Kingdom, and Sweden), tended to have relatively prompt access to all new antibiotics
The 2nd paper (and paper #3, the related editorial) focus on use of newer antibiotics in the US. As essentially all new antibiotics were approved in the US (and thus theoretically available), were they used? Analyzing data for the period Jan 2016 to Jun 2021, Strich et al. found limited use of safer, newer agents (ceftazidime–avibactam, ceftolozane–tazobactam, meropenem–vaborbactam, plazomicin, eravacycline, imipenem–relebactam–cilastatin, and cefiderocol). Specifically, 1091 of 2631 DTR infection episodes (41.5%) were treated exclusively using traditional agents. This included use of antibiotics such as polymyxins, aminoglycosides, and tigecycline in 865 of 1091 episodes (79.3%)..
——————————
Stop #3 (Impact of new agents — the new paper!): What is the impact of these newer agents on outcome in DTR infections?
Well, some use is better than none! So, are outcomes better when the newer agents are used? Here’s where we come to the new paper:
- Walker MK, Yek C, Sarzynski S, Warner S, Harris AD, Baghdadi JD, et al. Survival trends in patients with difficult-to-treat, antibiotic-resistant, Gram-negative infections in the era of next-generation antibiotics in the USA: a retrospective cohort study. The Lancet Infectious Diseases 2026. DOI: 10.1016/S1473-3099(26)00020-4.
- (and an editorial about the paper) Kabbani S, McDonald LC. Treatment of multidrug-resistant Gram-negative infections: A stewardship imperative. The Lancet Infectious Diseases 2026. DOI doi.org/10.1016/S1473-3099(26)00113-1.
Across an overlapping timeframe to that of the Strich 2024 study (1Jan2016 to 31Aug2023), Walker et al. retrieved electronic data on 8,319,398 adult inpatient encounters at 471 US hospitals. Across these inpatient events,
- There were 9,384 encounters (0.11%) with evidence of a DTR pathogen.
- Of these 9,384 encounters, the electronic data were consistent with a DTR infection in 5,065 (54.0%) of the encounters (rather than just colonization).
- NB: Pay close attention to the fact that overall # of DTR infections is small: only ~5,000 at ~500 hospitals (rounding a bit). That’s 1 DTR infection for every 1,643 inpatient encounters. We’ll need this later!
- Antibiotics used for these 5,065 DTR infections were broken down into 3 categories:
- New DTR-active antibiotics (cefiderocol, ceftazidime-avibactam, ceftolazane/tazobactam, eravacycline, imipenem/relebactam, meropenem/vaborbactam, omadacycline, and plazomicin)
- Traditional DTR-active antibiotics (amikacin, chloramphenicol, colistin, doxycycline, fosfomycin, gentamicin, minocycline, tetracycline, tigecycline, and tobramycin)
- Non-DTR-active antibiotics (everything else)
- Further, initial antibiotic therapy (therapy given on or before the day of the DTR culture) was classified as in vitro discordant if susceptibility data suggested the antibiotic (even a new DTR-active antibiotic) was inappropriate
So, let’s start by looking at physical availability of the new DTR-active antibiotics in the survey. As you’ll recall from the 1 Dec 2024 newsletter (“The 6 meanings of ‘Lack of Access’ (UNSLAP)”) and the related 7 Apr 2025 newsletter (“UNSLAP: You reach for the antibiotic … and it’s not there!”), if the antibiotic is not in your local pharmacy, it might as well not exist! The electronic data did not show what was actually on pharmacy shelves, but the authors were able to infer access by looking at use and susceptibility testing:
- In 2016-18, only 12% of hospitals reported use of a new DTR-active antibiotic and only 4% did susceptibility testing for the newer antibiotics.
- By 2021-23, the numbers had risen to 63% of hospitals using a newer antibiotic and 71% doing susceptibility testing.
OK, so far so good … a nice rise (but still only to about 2/3rds of hospitals). But, what therapy was actually given for those infections? Here’s where things go off the rails. See below Figure 3 from Walker et al.:
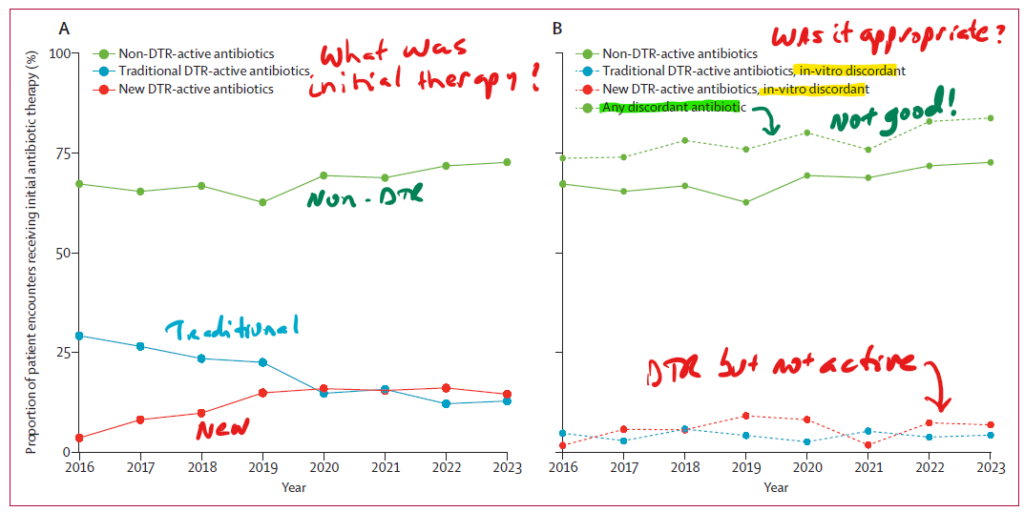
The x-axis is time (2016 to 2023) and the y-axis is % of DTR infections with the given type of initial antibiotic treatment. Critically, initial therapy is defined here as that given before the culture result is known — the therapy was empirical. We’ll look in a moment to see what happened when the culture result returns, but keep in mind that the initial period is critical to survival … we know from multiple studies that time to effective therapy makes a difference (as one example, see Liu XV et al., Amer J Resp Crit Care Med 2017, DOI: 10.1164/rccm.201609-1848oc)..
On the left of the figure, we have the raw data — a DTR-active antibiotic was initial therapy in only ~30% of DTR infections (total by eyeball of the red and blue lines at bottom). Ugh! But wait … it gets worse … when you further analyze (right-hand panel) the selected drug by whether it was actually active in vitro (that is, was there in vitro concordance or discordance), the authors found in vitro discordance in about half of the uses of a DTR antibiotic (total of the dotted red/blue lines at bottom). The net effect is is shown at the top of the right-hand panel … the percent of initial therapy predicted ineffective (either because the antibiotic used was non-DTR-active or was DTR-active but discordant) actually rose over the study period, peaking at 84% in 2023!
Now, it did turn out that therapy began to be changed as culture results were known. But, the delay in standard culture and susceptibility testing meant that those 5,065 patients were switched only slowly to a better DTR-active drug. We see that in this graphic of therapy by day relative to culture collection:
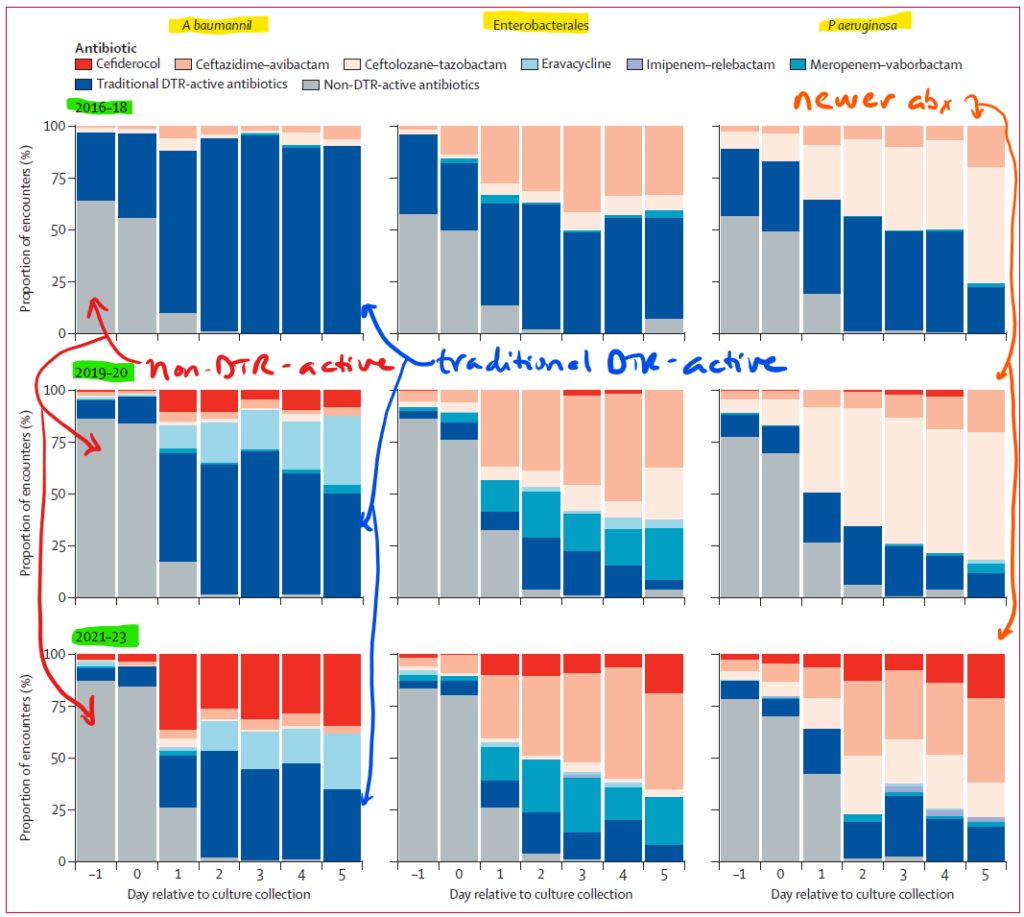
In the figure above (Figure 2 from Walker et al.), the x-axis is days relative to collection of the culture that yielded the DTR pathogen. Data by row are for 3 successive time periods (2016-18, 2019-20, 2021-23) and by column are for Acinetobacter baumannii, Enterobacterales, and Pseudomonas aeruginosa.
In the figure, now focus on the 3rd column from the left in each sub-figure … this is the time (1 day after culture collection) when new information might become available. You can see that therapy begins to change on that day … but that’s a long time to wait for effective therapy.
Well, at least some patients did get a potentially better therapy. So (cue drumroll), did better therapy make a difference for the population as a whole over the study period? Given that either a DTR antibiotic or (worse) an ineffective antibiotic was given in more than three quarters of the population, the effect of the newer antibiotics would be expected to be diluted out. Thus, you will be unsurprised to hear that the answer is no, a reduction in mortality was not seen. The overall estimated mortality probability for DTR infections was 20% for Enterobacterales, 20% for P. aeruginosa, and 25% for A. baumannii … and this really did not change over the study period. Aside: There was a hint of a reduction in patients with P. aeruginosa bloodstream infections, but the authors feel this is a weak observation (small N and hints in the data of unexplained confounding).
In discussing their data, the authors focus on two factors that could have obscured the benefits of newer therapies:
- Competing causes of mortality (age, other diseases) would obscure effects on the infection.
- Delay in appropriate therapy, especially in the subset (~25%) with sepsis.
And I would agree that both of these make sense … but I’d like to take this one step further and consider the amount of improvement that is even plausible given that there are multiple causes of mortality.
——————————
Stop #4 (One step further): How much of a mortality improvement is actually plausible?
Note: At this point in the newsletter, we have to assume that you are familiar with the study design ideas of superiority and non-inferiority. If not, please pause and (i) read the 19 Sep 2020 newsletter entitled “In Praise of Non-Inferiority” and (ii) watch its associated YouTube video.
We start in this analysis by looking at the overall estimated mortality probability for DTR infections: ~20% for Enterobacterales, ~20% for P. aeruginosa, and ~25% for A. baumannii.
So, if we’re going to find an improvement (a reduction in mortality), we have to find it in the 20-25% who died … the other 75-80% survived and we can’t further improve their outcomes.
And, let’s also recognize that some mortality is doubtless unrelated to the infection and thus unpreventable: this would be the idea of attributable vs. non-attributable mortality. It’s hard to know an exact figure, we but know from Table 1 of Walker et al. that 12-13% of patients in the survey had a do-not-resuscitate order … so perhaps we can use 10% as a conservative estimate for non-attributable mortality.
So now our zone for improvement (reduction) in mortality is limited to the attributable mortality subset … that’s a window from ~20% to (say) ~10%. And, it would take a 100% reduction in attributable mortality to reduce the mortality from 20% to 10%! That’s a tall order!
So, perhaps you are a bit less ambitious and think a better therapy could produce a reduction from 20% to 15% … effectively, a 50% relative reduction in the plausible zone for improvement. Well, that would be a stunning effect but it would take a study of about 1,250/arm to have 90% power to show that much change at P < 0.05. And if you thought you were going to show something a bit less (perhaps 40% relative reduction, the amount of mortality increase suggested by Kadri 2018), you’d need ~2,000/arm for a change from 20% to 16%. You could reduce the sample size to ~1,600/arm by accepting a P < 0.10 for your design, but that’s still a big study!
Ouch! But, you say, “I thought superiority studies were supposed to be small!” Well, that’s true for a really big effect — you only need 63/arm to show a change from 65% to 35% — but here we’re dealing with much smaller changes. Even if you said you’d recover all the mortality by moving the needle from 20% mortality to 10%, you’d need 286/arm. Not vast but not a small study either.
——————————
Stop #5 (Implications, Part 1): Can we ever show that new drugs are better?
So, let’s return to the opening statements. Deep, deep down, this conversation is about how we know whether (or not) new drugs really work for bad bugs. Yes, we can see the favorable in vitro microbiology. Yes, we can see the favorable results of animal models. But, what about in human infections? “I want to see data on new drugs for bad bugs in people,” you cry! “Why are you bringing me these confusing non-inferiority results? Why aren’t you studying bad bugs? And, superiority studies would be cleaner! They can be smaller! Why aren’t we instead seeing superiority data on treatment of bad infections?”
Hopefully, the answers to these questions are now clear:
- Unlike animal model studies, we can’t directly show the benefit of a new therapy over no treatment (placebo) in human studies because we can’t deliberately do placebo-controlled studies of new therapies … we always seek active therapy in all study arms.
- Infections can be fatal if not treated promptly! Placebo just is not an option!
- 25 Apr 2026 addendum: This was extensively debated 2007-08 (see Spellberg et al. Clin Investig (Lond). 2011;1:19-32; https://doi.org/10.4155/cli.10.1 for a good summary). Ultimately, FDA explicitly stated that placebo was not ethical in its guidance document for community-acquired bacterial pneumonia.
- Situations where superiority studies have the potential to succeed are rare because we work hard to control the rate of DTR, MDR, XDR, and PDR pathogens.
- We do sometimes run studies in which highly resistant pathogens are sought but these studies tend to be smaller and hard to enroll because we work hard to make resistant pathogens rare and thus …
- … Walker et al. needed data from 471 hospitals spanning 8 years to find the 5,000 DTR infections in their report! That size and duration is obviously not feasible for a drug development program.
- Finally, superiority studies are most feasible when there is a VERY large effect to be measured. Small effects require huge studies … and small changes are often not very compelling even when shown.
- Now, you would correctly argue that perhaps we’re using the wrong endpoint … mortality is a blunt measure. Would measures that incorporate a range of non-mortality clinical measures be helpful?
- This is the idea, for example, of DOOR (Desirability Of Outcome Rating) that has been championed over the years by Scott Evans and colleagues. Yes, this could be a helpful path forward, especially if those endpoints can be shown to be clinical relevant and if superiority can be shown on a non-mortality component of an endpoint.
- For an introduction to this literature, see Evans SR et al. Desirability of Outcome Ranking (DOOR) and Response Adjusted for Duration of Antibiotic Risk (RADAR), Clinical Infectious Diseases, 61:800–806, 2015, https://doi.org/10.1093/cid/civ495.
- But, mortality is such an obvious and powerful endpoint that it’s hard to get away from it, especially in settings like infection where the course of the illness is often rapid and obvious. Even though it can be argued that other endpoints are clinically relevant and potentially more sensitive, the fundamental data that justify many of our standard designs depend on ACM as the primary endpoint because this is the measure that can be used to justify a non-inferiority margin.
And so, this is why we use non-inferiority studies:
- We know from non-clinical data that the new antibiotic could work.
- We show in a UDR setting that the new agent is non-inferior to an existing agent when the existing agent still works.
- When resistance arises to the existing agent, the new agent’s activity is unaffected … it still works.
- We can supplement the non-inferiority study with salvage studies in patients with MDR, XDR, PDR, or DTR infections but these studies are (hopefully) almost impossible to enroll if we are doing a good job with infection control.
- The key to approval lies in the non-inferiority study done in the UDR setting
- And, this is what allows us to develop new agents before the need for them becomes acute!
- For more on this critical theme, see this YouTube video explainer or the 19 Sep 2020 newsletter entitled “In praise of non-inferiority.”
——————————
Stop #5 (Implications, Part 2): What does this mean for calls to improve outcomes through alternative approaches such as host-directed therapies?
Well, you should by now be realizing that the news is not great. Unless your approach has a simply stunning effect, the ability to show a benefit in clinical studies is very limited. We say this with sadness as we think many of those ideas make good scientific sense. Unfortunately, it is very, very hard to show such effects consistently unless the effect is dramatic. For more on this (really, we have got to get to the end here!!), please see the 6 Aug 2019 newsletter entitled “Non-traditional antibiotics: A pipeline review and an analysis of key development challenges”. It’s a relatively old newsletter, but it’s one that covered in depth the problem of developing non-traditional therapies (such as host-directed therapies) and one where new material has been added as it has arisen.
——————————
(Implications, Part 3) Rapid diagnostics and access to DTR-active agents are the keys to taking action!
So, there two strong, clear action items that come from Walker et al.: The first is to make a diagnosis, and make it quickly! This one is quite obvious …if antibiotics are the #FireExtinguishersOfMedicine, then diagnostics are surely the #SmokeDetectorsOfMedicine! But as noted in the paper, “Diagnosis of a DTR pathogen by conventional antimicrobial susceptibility testing methods is estimated to take a mean of 92.2 hours (SD 27.8) and can result in a significant delay in the time to initiation of an appropriate antibiotic.” That’s 4 days — much too long! Quoting further from Walker et al.:
- “…rapid diagnostics could enable earlier yet stewardly initiation of newer over traditional antibiotics in this patient population with a high burden of sepsis.
- “However, current rapid diagnostic platforms focus on enzyme-mediated resistance mechanisms, such as Klebsiella pneumoniae carbapenemases or New Delhi metallo-β-lactamases, despite the fact that such mechanisms are present in only a small fraction of resistant isolates and DTR isolates often have nonenzyme-mediated resistance mechanisms.
- “Empirical antibiotic therapy could also be guided by predictive algorithms that estimate the likelihood of resistant pathogens, but this approach warrants further investigation.”
Is “stewardly” really a word? Well, it works for us and the message is clear. Also clear is the comment about the limitations on current diagnostics — we need tools for detection of more than just beta-lactamases! In further support of the #SmokeDetectorsOfMedicine, see these recent newsletters on the power of diagnostics:
- 18 June 2025 newsletter: “Extending STEDI to diagnostics: STRIDES”
- 17 Feb 2026 newsletter: “Workshop on diagnostics: Can we drive use by making value visible?”
And the second is, of course, ensure your healthcare system has access to all the DTR-active agents, especially the newer ones! Here again we remind you of the 1 Dec 2024 newsletter (“The 6 meanings of ‘Lack of Access’ (UNSLAP)” and the related 7 Apr 2025 newsletter (“UNSLAP: You reach for the antibiotic … and it’s not there!”). The titles of those newsletters are self-explanatory: if the antibiotic is not in your local pharmacy, it might as well not exist!
——————————
Whew! That was a lot! Apologies for going on such a long tour, but it seemed helpful. Thanks for listening! All best wishes, John & Aaron
John H. Rex, MD | Chief Medical Officer, F2G Ltd. | Operating Partner, Advent Life Sciences. Follow me on Twitter: @JohnRex_NewAbx. See past newsletters and subscribe for the future: https://amr.solutions/blog/. All opinions are my own.
Aaron Dane, CStat | DaneStat Consulting Limited | aarondane@danestat.com | www.linkedin.com/in/aaronldane | All opinions are my own.
Summary of prior newsletters on DTR and key related topics:
- 13 Jan 2019: “Categories of resistance: MDR, XDR, PDR, UDR, and (new!) DTR”
- 20 Feb 2020: “Language matters: CRE vs. CPE; SDD vs. I; and MDR, XDR, PDR, UDR vs. DTR”
- 7 Jun 2020: “Assessing antibiotic value: DTR, fire extinguishers, and a view from Australia”
- 19 Sep 2020: “In praise of non-inferiority”
- 17 Mar 2021: “Where’s the innovation? / Indian Priority Pathogen List / DTR video explainer”
- UDR vs. MDR/XDR YouTube video explainer — see below!
- 25 Apr 2025: “Evolving the idea of DTR (Difficult-to-Treat Resistance) to include antibiotic access”
John’s Top Recurring Meetings
Virtual meetings are easy to attend, but regular attendance at annual in-person events is the key to building your network and gaining deeper insight. My personal favorites for such in-person meetings are below. Of particular value for developers, the small meeting format of BEAM’s AMR Conference (March) and GAMRIC (September-October; formerly, the ESCMID-ASM conference series) creates excellent global networking. IDWeek (October) and ECCMID (April) are much larger meetings but also provide opportunities for networking with a substantial, focused audience via their Pipeline sessions. Hope to see you there!
- 22-24 Sep 2026 (Lisbon, Portugal): The 2nd GAMRIC, the Global AMR Innovators Conference (London, UK). Formerly the ESCMID-ASM (or ASM-ESCMID depending on location) Joint Conference on Drug Development for AMR, 2026 will be the 11th year for this series that is now under the joint sponsorship of CARB-X, ESCMID, BEAM Alliance, GARDP, LifeArc, Boston University, and AMR.Solutions. The ongoing series employs the successful format of prior meetings with a single-track meeting and substantial networking time. The 2025 meeting was a sell-out success! A written summary of the 2025 meeting is here and the videos from the sessions are now available here. Registration for the 2026 meeting is now open and you can view the preliminary program here. Abstract submissions are open through Fri 10 Apr at noon CET; note as well that travel grants are available for attendees with accepted abstracts. A late-breaker window be announced for July.
- 21-24 Oct 2026 (Washington, DC, USA): IDWeek 2026, the annual meeting of the Infectious Diseases Society of America. I would expect the program to continue to provide a substantial opportunity to present a product to a large audience (see also adjacent note about ESCMID) as well as opportunities to present at an IDWeek Pipeline Session.
- 23-24 Mar 2027 (Basel, Switzerland): The 10th AMR Conference (3-4 Mar 2026) is now over and offered a rich program that included a 10-year retrospective (we’ve done a lot!), regulatory updates, discussions of how to pursue development in China, and much more … in addition to being a superb opportunity for networking! I am told the session videos will soon be available on the conference website. Mark your calendars for next year’s 11th AMR Conference!
- 9-13 April 2027 (Stockholm, Sweden): ESCMID Global 2027, the annual meeting of the European Society for Clinical Microbiology and Infectious Diseases. Details won’t be out for some months, but the website is here. I do know that the meeting schedule will again include a Science Policy Forum on Friday 9 April 2027 (see here the newsletter about the 2026 science policy forum) and all who are interested in Pull incentives and the antibiotic ecosystem should plan to be there!)..
Upcoming meetings of interest to the AMR community:
- 28 Apr 2026 (9:30-11:00 CET, virtual): REVIVE (GARDP)-sponsored webinar: “Natural product-inspired antibiotics: Successes and future prospects.” Go here for details and to register.
- 4-8 June 2026 (Washington, DC): ASM Microbe, the annual meeting of the American Society for Microbiology. The meeting format is evolving and next year will combine 3 meetings (ASM Health, ASM Applied and Environmental Microbiology, and ASM Mechanism Discovery) into one event. Go here for details.
- 11-12 Jun 2026 (Washington, DC): The Second Annual Unite for Sepsis Symposium, presented by the Sepsis Alliance. The event seeks to accelerate progress in sepsis research and care. Go here for details and to register.
- 22-24 Sep 2026 GAMRIC (Lisbon, Portugal), the Global AMR Innovators Conference (London, UK; formerly the ESCMID-ASM Joint Conference on Drug Development for AMR). See list of Top Recurring meetings, above..
- 10-18 Oct 2026 (Annecy, France, residential in-person program): ICARe (Interdisciplinary Course on Antibiotics and Resistance) … and 2026 will be the 10th year for this program. Patrice Courvalin orchestrates content with the support of an all-star scientific committee and faculty. The resulting soup-to-nuts training covers all aspects of antimicrobials, is very intense, and routinely gets rave reviews! Registration for 2026 will be during March 16–July 3, 2026. You can go here for more details and you should put a reminder in your calendar register on / after 16 March.
- 21-24 Oct 2026 (Washington, DC, USA): IDWeek 2026. See list of Top Recurring meetings, above.
- 10-13 November 2026 (Madrid, Spain): The International Society for Infectious Diseases (ISID) has announced its 21st International Congress on Infectious Diseases (ICID). Register and view the preliminary program here (Early bird closes 30 July 2026); abstract deadline is 28 April 2026.
- 23-24 Mar 2027 (Basel, Switzerland): The 11th AMR Conference sponsored by the BEAM Alliance. See list of Top Recurring meetings, above.
- 9-13 April 2027 (Stockholm, Sweden): ESCMID Global 2026, the annual meeting of the European Society for Clinical Microbiology and Infectious Diseases. See Recurring Meetings list, above.
- ??? Mar 2028 (yes, that’s 2028, with location TBD): The 2028 Gordon Research Conference (GRC, https://www.grc.org/) entitled “Antibacterials of Tomorrow to Combat the Global Threat of Antimicrobial Resistance” and its related Gordon Research Seminar (GRS) for young doctoral and post-doctoral researchers will be sometime in March 2028. The organizers hope to coordinate dates and location with the 2028 BEAM-AMR meeting. Details to follow — mark your calendar!
Self-paced courses, online training materials, and other reference materials:
- OpenWHO: “Antimicrobial Resistance in the environment: key concepts and interventions.” Per the webpage for the course, it will teach you “…why addressing AMR in the environment is essential and gain insights into how action can be taken to prevent and control AMR in the environment at the national level.” This course builds on WHO’s 2024 Guidance on wastewater and solid waste management for manufacturing of antibiotics. For further reading, see also the 25 Sep 2023 newsletter entitled “Manufacturing underpins both access and stewardship: Cefiderocol as a case study” and the 28 Jan 2024 newsletter entitled “EMA Concept Paper: Guidance on manufacturing of phage products”.
- GARDP’s REVIVE website provides an encyclopedia covering a range of R&D terms, recordings of prior GARDP webinars, a variety of viewpoint articles, and more! Check it out!
- GARDP’s https://antibioticdb.com/ is an open-access database of antibacterial agents.
- The CARB-X website provides a range of recordings from its webinars, bootcamps, and more. A bit of browsing would be time well spent!
- British Society for Antimicrobial Chemotherapy offers an eLearning section: Education – The British Society for Antimicrobial Chemotherapy.
- [NEW] Wellcome Leap have announced a $50m funding round to support development of technologies that would spare the gut, focus the delivery of the antibiotic to the site of infection, and (thereby) reduce the rate of emergence of resistance. The application timeline is soon and short: 8-15 May 2026. See the 19 Apr 2026 newsletter for details.
- CARB-X have announced a funding round that will be open 8-22 April 2026 (there will also be a round during 4Q 2026). There are 4 funding themes as below. Informational webinars have been announced for 9 and 10 April 2026.
- Direct-acting therapeutics for infections caused by Gram-negative bacteria
- Novel Chemistry for AMR Challenge – target-based therapeutics.
- Non-vaccine approaches to prevent neonatal sepsis
- Diagnostics for neonatal sepsis
- The Horizon Europe Work Programme 2026-2027 includes at least 3 calls of interest within its Cluster 1 — see the list below. The application window starts 10 Feb 2026 and closes on 16 Apr 2026. See also the 12 Dec 2025 newsletter about the call. Note as well that there calls for agents to prevent and/or treat viral infections.
- HORIZON-HLTH-2027-01-DISEASE-08: Development of innovative antimicrobials against pathogens resistant to antimicrobials
- HORIZON-HLTH-2027-02-IND-02: Portable point-of-care diagnostics
- HORIZON-HLTH-2026-01-DISEASE-03:Advancing research on the prevention, diagnosis, and management of post-infection long-term conditions.
- ENABLE-2 has continuously open calls for both its Hit-to-Lead program as well as its Hit Identification/Validation incubator. Applicants must be academics and non-profits in Europe due to restrictions from the funders. Applications are evaluated in cycles … see the website for details on current timing for reviews.
- BARDA’s long-running BAA (Broad Agency Announcement) for medical countermeasures (MCMs) for chemical, biological, radiological, and nuclear (CBRN) threats, pandemic influenza, and emerging infectious diseases is now BAA-23-100-SOL-00004 and offers support for both antibacterial and antifungal agents (as well as antivirals, antitoxins, diagnostics, and more). Note especially these Areas of Interest: Area 3.1 (MDR Bacteria and Biothreat Pathogens), Area 3.2 (MDR Fungal Infections), and Area 7.2 (Antibiotic Resistance Diagnostics for Priority Bacterial Pathogens). Although prior BAAs used a rolling cycle of 4 deadlines/year, the updated BAA released 26 Sep 2023 has a 5-year application period that ends 25 Sep 2028 and is open to applicants regardless of location: BARDA seeks the best science from anywhere in the world! See also this newsletter for further comments on the BAA and its areas of interest.
- HERA Invest was launched August 2023 with €100 million to support innovative EU-based SMEs in the early and late phases of clinical trials. Part of the InvestEU program supporting sustainable investment, innovation, and job creation in Europe, HERA Invest is open for application to companies developing medical countermeasures that address one of the following cross-border health threats: (i) Pathogens with pandemic or epidemic potential, (ii) Chemical, biological, radiological and nuclear (CBRN) threats originating from accidental or deliberate release, and (iii) Antimicrobial resistance (AMR). Non-dilutive venture loans covering up to 50% of investment costs are available. A closing date is not posted insofar as I can see — applications are accepted on a rolling basis; go here for more details.
- The AMR Action Fund is open on an ongoing basis to proposals for funding of Phase 2 / Phase 3 antibacterial therapeutics. Per its charter, the fund prioritizes investment in treatments that address a pathogen prioritized by the WHO, the CDC and/or other public health entities that: (i) are novel (e.g., absence of known cross-resistance, novel targets, new chemical classes, or new mechanisms of action); and/or (ii) have significant differentiated clinical utility (e.g., differentiated innovation that provides clinical value versus standard of care to prescribers and patients, such as safety/tolerability, oral formulation, different spectrum of activity); and (iii) reduce patient mortality. It is also expected that such agents would have the potential to strongly address the likely requirements for delinked Pull incentives such as the UK (NHS England) subscription pilot and the PASTEUR Act in the US. Submit queries to contact@amractionfund.com.
- INCATE (Incubator for Antibacterial Therapies in Europe) is an early-stage funding vehicle supporting innovation vs. drug-resistant bacterial infections. The fund provides advice, community, and non-dilutive funding (€10k in Stage I and up to €250k in Stage II) to support early-stage ventures in creating the evidence and building the team needed to get next-level funding. Details and contacts on their website (https://www.incate.net/).
- These things aren’t sources of funds but would help you develop funding applications
- The Global AMR R&D Hub’s dynamic dashboard (link) summarizes the global clinical development pipeline, incentives for AMR R&D, and investors/investments in AMR R&D. See also the 7 Feb 2026 newsletter (“The global funding pipeline, 2017-2023: A review”) about an excellent deep dive by the Hub team into patterns of funding over time.
- Antimicrobial Resistance Research and Innovation in Australia is an actively updated summary that covers Australia’s AMR research and patent landscape. It is provided via collaboration between The Lens (an ambitious project seeking to discover, analyse, and map global innovation knowledge) and CSIRO (Commonwealth Scientific and Industrial Research Organisation, an Australian Government agency responsible for scientific research). Lots to explore here!
- Diagnostic developers would find valuable guidance in this 6-part series on in vitro diagnostic (IVD) development. Sponsored by CARB-X, C-CAMP, and FIND, it pulls together real-life insights into a succinct set of tutorials.
- In addition to the lists provided by the Global AMR R&D Hub, you might also be interested in my most current lists of R&D incentives (link) and priority pathogens (link).