Dear All:
ESCMID Global once again held an afternoon of sessions focused on the intersection of AMR-related science and policy. With an overall theme of “Smart policies for lean times,” the well-attended afternoon comprised a pair of workshops, a plenary session, and a networking reception (I’ll also note that there was a related integrated workshop [“Aligning research and policy to unlock antibiotic innovation”, supported by GSK] that preceded the afternoon but I was unable to attend it).
Importantly, I am told that this series is going to continue to be implemented on the Friday that begins ESCMID. Next year, that will be Friday 9 Apr 2027 with ESCMID running 9-13 April 2027 in Stockholm. So, mark your calendars now for next year!
Back to this year, the afternoon’s parallel workshops were evidence-focused:
- Evidence for policy: modelling and economics
- Innovation for action: diagnostics and cost-effectiveness across the product development and implementation pathway
Both session were full to overflowing with standing room only for later arrivals!

Above, photo credit Robert Skov — lots of interest in these workshops … we need bigger rooms next year!
After the workshops, the plenary (opened by Robert Skov and then chaired by Jon Friedland) featured:
- (opening comments) Hon. Minister Mekdes Daba, Global Leaders Group on AMR, and Tedros Adhanom Ghebreyesus, WHO Director-General
- (keynote) “AMR policy action under pressure: doing more with less”: Naomi Rupasinghe (World Bank, Washington, USA)
- “Efficient and effective IPC [Infection Prevention and Control]”: Benedetta Allegranzi (WHO Regional Office for The Eastern Mediterranean, Cairo, Egypt)
- “Prevention pays off: vaccination policies for AMR” by Padmini Srikantiah (Gates Foundation, Seattle, USA)
- “Global AWaRe targets and national cost savings“: Koen Pouwels (University of Oxford, Oxford, UK)
- “A gram of prevention or a kilo of cure?“: Javier Yugueros-Marcos (World Organisation for Animal Health, Paris, France)
A full summary of the afternoon’s content is more than I can make happen lest this newsletter become a version of a long novel! But, I would like to give a tour of the ideas shared by each speaker:
First up, Naomi Rupasinghe’s keynote focused on the ways that she has been working from the World Bank to create workable investment opportunities in AMR. I’ve gotten to know her work through sitting with her on the World Economic Forum’s AMR Global Future Council and its 2026 Davos Compact on AMR that now has > 50 signatories (see this blog).
Now, you may well react to “investment” and “AMR” with “no one wants to go there”, but there are examples of creating impact or social investment possibilities for investors who want to assist social causes such as health care, housing or education. A 22 Sep 2019 Wall Street Journal story entitled “Bonds That Do Good—and, Maybe, Well” provides a good summary of these ideas as does the discussion in the 11 Nov 2025 newsletter entitled (“Milken Future of Health Summit: AMR Symposium”).
Naomi’s talk showed us the path forward in AMR with an example from the vaccine world where the IFFIm (International Finance Facility for Immunisation) leveraged long-term pledges from 11 sovereign governments to raise $6.2b from the capital markets … and thus, 233m children were immunized:

For more on this type of work see this UNESCO report entitled “Advanced Market Commitment [AMC] for Pneumococcal Vaccines” as well as this pair of reports from GAVI about the pneumococcal vaccine AMC and its impact
Next up, WHO’s Benedetta Allegranzi summarized the progress we’ve made in developing (and implementing) best practices for IPC (Infection Prevention and Control). In 2023, we had release of the WHO’s Global Strategy on IPC and then in 2025 we had the release of a practical guide to Development and implementation of national action plans for infection prevention and control: practical guide:

The third speaker, Padmini Srikantiah, Deputy Director at the Gates Foundation, gave us a tour of the impact of vaccines. She noted that existing vaccines (alone!) would annually avert 106k deaths, 9.1m DALYs, US$861m in hospital costs, and US$5.9b in productivity losses. Combined with future vaccines, the potential value is staggering!
I particularly enjoyed this figure highlighting the durability of vaccines. The x-axis is time (years) and each line represents a drug or vaccine, starting from the time of its introduction to the first documented resistance-related failure in a human (an X). Look closely! All the red drug lines end in an X whereas only a few of the vaccines end with an X … and even then, it takes many years for resistance to develop to vaccines vs. often relatively prompt resistance to the drugs.
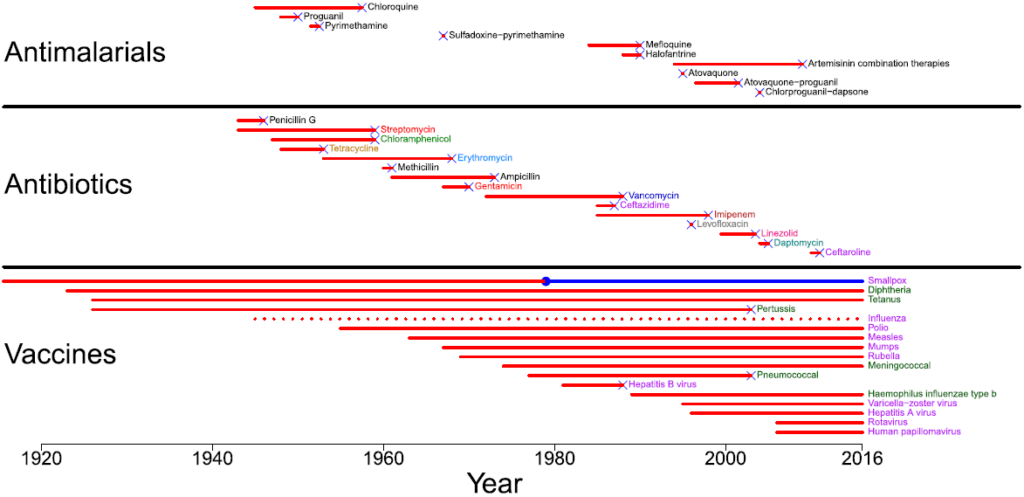
Figure above is from Kennedy DA and Read AF. Why the evolution of vaccine resistance is less of a concern than the evolution of drug resistance. Proceedings of the National Academy of Sciences. 2018;115(51):12878–86, https://doi.org/10.1073/pnas.1717159115.
Fourth, Koen Pouwels (U Oxford), reviewed his research on the value of adjusting prescribing patterns so that usage in the Access category of the WHO AWaRe (Access-Watch-Reserve) antibiotic ranking hits the UNGA HLM-AMR target of ~70%. Based on modeling that grouped regions by sociodemographic factors and infection patterns (“latent class modeling”), substantial savings were possible … and please read his paper for more details! And for more on AWaRe, see the 13 Dec 2022 newsletter entitled “WHO AWaRe antibiotic handbook released!”

Fifth, Javier Yugueros-Marcos (World Organisation for Animal Health) reminded us that AMR is already causing $17b in annual GDP losses in livestock production with the potential for the cost to rise to $159b annually by 2050 if we don’t act (26 Sep 2024 report from WOAH). Building on a classic concept with his title “A gram of prevention or a kilo of cure,” he showed the potential for modern interventions to have a huge impact (in this example using AI-enabled CCTV monitoring):

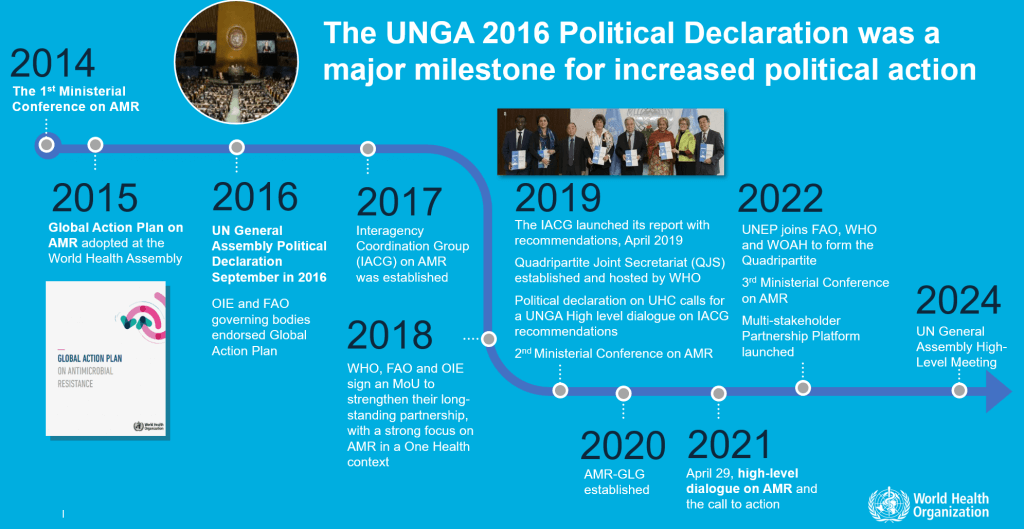
Finally, WHO’s JP Nyemazi closed the session by first reviewing the political work that brought us to the 2024 UNGA HLM on AMR (graphic from 15 Apr 2023 newsletter):
… followed by a discussion of what is coming for IPEA (the Independent Panel on Evidence for Action; see the 6 Jul 2025 newsletter entitled “Establishing an Independent Panel on Evidence for Action”) as well as updates to the Global Action Plan (GAP) on AMR:
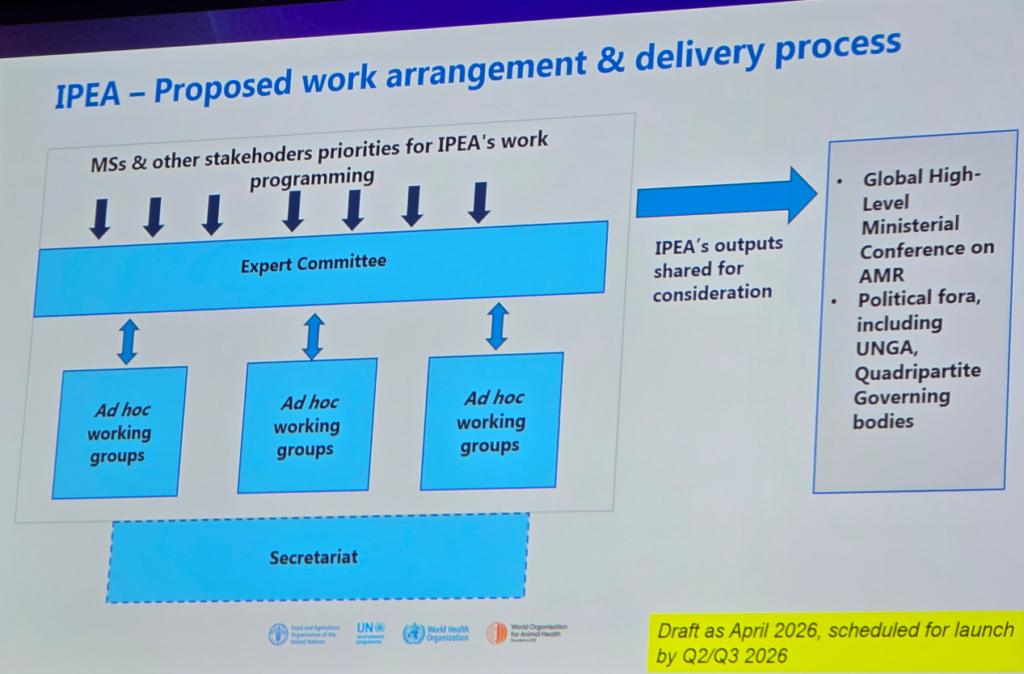

As noted on the slides, the IPEA is scheduled to launch by 3Q26; the updated GAP will be endorsed next month at the World Health Assembly (WHA).
Whoosh! Very exciting to see all this work underway! We truly do need “Smart policies for lean times” so that we effectively deploy the resources available. By continuing to work in a collaborative way across OneHealth sectors, we can make the case to policy makers for thoughtful, integrated solutions that are sustainable over generations.
What a great afternoon … many thanks to ESCMID for this ongoing series! And after all of that, we were ready for a beer at the networking reception! If you were there, it was great to see you! If you weren’t there … we’ll see everyone at this event next year!
All best wishes, –jr
John H. Rex, MD | Chief Medical Officer, F2G Ltd. | Operating Partner, Advent Life Sciences. Follow me on Twitter: @JohnRex_NewAbx. See past newsletters and subscribe for the future: https://amr.solutions/blog/. All opinions are my own.
John’s Top Recurring Meetings
Virtual meetings are easy to attend, but regular attendance at annual in-person events is the key to building your network and gaining deeper insight. My personal favorites for such in-person meetings are below. Of particular value for developers, the small meeting format of BEAM’s AMR Conference (March) and GAMRIC (September-October; formerly, the ESCMID-ASM conference series) creates excellent global networking. IDWeek (October) and ECCMID (April) are much larger meetings but also provide opportunities for networking with a substantial, focused audience via their Pipeline sessions. Hope to see you there!
- 17-21 April 2026 (Munich, Germany): ESCMID Global 2026, the annual meeting of the European Society for Clinical Microbiology and Infectious Diseases. You can go here to register and view the program.The meeting schedule will again includes a Science Policy Forum on Friday afternoon (all who are interested in Pull incentives should be there!) and an innovation-focused Pipeline Monday.
- 22-24 Sep 2026 (Lisbon, Portugal): The 2nd GAMRIC, the Global AMR Innovators Conference (London, UK). Formerly the ESCMID-ASM (or ASM-ESCMID depending on location) Joint Conference on Drug Development for AMR, 2026 will be the 11th year for this series that is now under the joint sponsorship of CARB-X, ESCMID, BEAM Alliance, GARDP, LifeArc, Boston University, and AMR.Solutions. The ongoing series employs the successful format of prior meetings with a single-track meeting and substantial networking time. The 2025 meeting was a sell-out success! A written summary of the 2025 meeting is here and the videos from the sessions are now available here. Registration for the 2026 meeting is now open and you can view the preliminary program here. Abstract submissions are open through Fri 10 Apr at noon CET; note as well that travel grants are available for attendees with accepted abstracts. A late-breaker window be announced for July.
- 21-24 Oct 2026 (Washington, DC, USA): IDWeek 2026, the annual meeting of the Infectious Diseases Society of America. Details are not yet available but I would expect the program to continue to provide a substantial opportunity to present a product to a large audience (see also adjacent note about ESCMID) as well as opportunities to present at an IDWeek Pipeline Session.
- 23-24 Mar 2027 (Basel, Switzerland): The 10th AMR Conference (3-4 Mar 2026) is now over and offered a rich program that included a 10-year retrospective (we’ve done a lot!), regulatory updates, discussions of how to pursue development in China, and much more … in addition to being a superb opportunity for networking! I am told the session videos will soon be available on the conference website. Mark your calendars for next year’s 11th AMR Conference!
Upcoming meetings of interest to the AMR community:
- 17-21 April 2026 (Munich, Germany): ESCMID Global 2026, the annual meeting of the European Society for Clinical Microbiology and Infectious Diseases. See Recurring Meetings list, above.
- 28 Apr 2026 (9:30-11:00 CET, virtual): REVIVE (GARDP)-sponsored webinar: “Natural product-inspired antibiotics: Successes and future prospects.” Go here for details and to register.
- 4-8 June 2026 (Washington, DC): ASM Microbe, the annual meeting of the American Society for Microbiology. The meeting format is evolving and next year will combine 3 meetings (ASM Health, ASM Applied and Environmental Microbiology, and ASM Mechanism Discovery) into one event. Go here for details.
- 11-12 Jun 2026 (Washington, DC): The Second Annual Unite for Sepsis Symposium, presented by the Sepsis Alliance. The event seeks to accelerate progress in sepsis research and care. Go here for details and to register.
- 22-24 Sep 2026 GAMRIC (Lisbon, Portugal), the Global AMR Innovators Conference (London, UK; formerly the ESCMID-ASM Joint Conference on Drug Development for AMR). See list of Top Recurring meetings, above..
- 10-18 Oct 2026 (Annecy, France, residential in-person program): ICARe (Interdisciplinary Course on Antibiotics and Resistance) … and 2026 will be the 10th year for this program. Patrice Courvalin orchestrates content with the support of an all-star scientific committee and faculty. The resulting soup-to-nuts training covers all aspects of antimicrobials, is very intense, and routinely gets rave reviews! Registration for 2026 will be during March 16–July 3, 2026. You can go here for more details and you should put a reminder in your calendar register on / after 16 March.
- 21-24 Oct 2026 (Washington, DC, USA): IDWeek 2026. See list of Top Recurring meetings, above.
- [Abstract deadline is 28 April!] 10-13 November 2026 (Madrid, Spain): The International Society for Infectious Diseases (ISID) has announced its 21st International Congress on Infectious Diseases (ICID). Register and view the preliminary program here (Early bird closes 30 July 2026); abstract deadline is 28 April 2026.
- 23-24 Mar 2027 (Basel, Switzerland): The 11th AMR Conference sponsored by the BEAM Alliance. See list of Top Recurring meetings, above.
- ??? Mar 2028 (yes, that’s 2028, with location TBD): The 2028 Gordon Research Conference (GRC, https://www.grc.org/) entitled “Antibacterials of Tomorrow to Combat the Global Threat of Antimicrobial Resistance” and its related Gordon Research Seminar (GRS) for young doctoral and post-doctoral researchers will be sometime in March 2028. The organizers hope to coordinate dates and location with the 2028 BEAM-AMR meeting. Details to follow — mark your calendar!
Self-paced courses, online training materials, and other reference materials:
- OpenWHO: “Antimicrobial Resistance in the environment: key concepts and interventions.” Per the webpage for the course, it will teach you “…why addressing AMR in the environment is essential and gain insights into how action can be taken to prevent and control AMR in the environment at the national level.” This course builds on WHO’s 2024 Guidance on wastewater and solid waste management for manufacturing of antibiotics. For further reading, see also the 25 Sep 2023 newsletter entitled “Manufacturing underpins both access and stewardship: Cefiderocol as a case study” and the 28 Jan 2024 newsletter entitled “EMA Concept Paper: Guidance on manufacturing of phage products”.
- GARDP’s REVIVE website provides an encyclopedia covering a range of R&D terms, recordings of prior GARDP webinars, a variety of viewpoint articles, and more! Check it out!
- GARDP’s https://antibioticdb.com/ is an open-access database of antibacterial agents.
- The CARB-X website provides a range of recordings from its webinars, bootcamps, and more. A bit of browsing would be time well spent!
- British Society for Antimicrobial Chemotherapy offers an eLearning section: Education – The British Society for Antimicrobial Chemotherapy.
- [NEW] Wellcome Leap have announced a $50m funding round to support development of technologies that would spare the gut, focus the delivery of the antibiotic to the site of infection, and (thereby) reduce the rate of emergence of resistance. The application timeline is soon and short: 8-15 May 2026. See the 19 Apr 2026 newsletter for details.
- CARB-X have announced a funding round that will be open 8-22 April 2026 (there will also be a round during 4Q 2026). There are 4 funding themes as below. Informational webinars have been announced for 9 and 10 April 2026.
- Direct-acting therapeutics for infections caused by Gram-negative bacteria
- Novel Chemistry for AMR Challenge – target-based therapeutics.
- Non-vaccine approaches to prevent neonatal sepsis
- Diagnostics for neonatal sepsis
- The Horizon Europe Work Programme 2026-2027 includes at least 3 calls of interest within its Cluster 1 — see the list below. The application window starts 10 Feb 2026 and closes on 16 Apr 2026. See also the 12 Dec 2025 newsletter about the call. Note as well that there calls for agents to prevent and/or treat viral infections.
- HORIZON-HLTH-2027-01-DISEASE-08: Development of innovative antimicrobials against pathogens resistant to antimicrobials
- HORIZON-HLTH-2027-02-IND-02: Portable point-of-care diagnostics
- HORIZON-HLTH-2026-01-DISEASE-03:Advancing research on the prevention, diagnosis, and management of post-infection long-term conditions.
- ENABLE-2 has continuously open calls for both its Hit-to-Lead program as well as its Hit Identification/Validation incubator. Applicants must be academics and non-profits in Europe due to restrictions from the funders. Applications are evaluated in cycles … see the website for details on current timing for reviews.
- BARDA’s long-running BAA (Broad Agency Announcement) for medical countermeasures (MCMs) for chemical, biological, radiological, and nuclear (CBRN) threats, pandemic influenza, and emerging infectious diseases is now BAA-23-100-SOL-00004 and offers support for both antibacterial and antifungal agents (as well as antivirals, antitoxins, diagnostics, and more). Note especially these Areas of Interest: Area 3.1 (MDR Bacteria and Biothreat Pathogens), Area 3.2 (MDR Fungal Infections), and Area 7.2 (Antibiotic Resistance Diagnostics for Priority Bacterial Pathogens). Although prior BAAs used a rolling cycle of 4 deadlines/year, the updated BAA released 26 Sep 2023 has a 5-year application period that ends 25 Sep 2028 and is open to applicants regardless of location: BARDA seeks the best science from anywhere in the world! See also this newsletter for further comments on the BAA and its areas of interest.
- HERA Invest was launched August 2023 with €100 million to support innovative EU-based SMEs in the early and late phases of clinical trials. Part of the InvestEU program supporting sustainable investment, innovation, and job creation in Europe, HERA Invest is open for application to companies developing medical countermeasures that address one of the following cross-border health threats: (i) Pathogens with pandemic or epidemic potential, (ii) Chemical, biological, radiological and nuclear (CBRN) threats originating from accidental or deliberate release, and (iii) Antimicrobial resistance (AMR). Non-dilutive venture loans covering up to 50% of investment costs are available. A closing date is not posted insofar as I can see — applications are accepted on a rolling basis; go here for more details.
- The AMR Action Fund is open on an ongoing basis to proposals for funding of Phase 2 / Phase 3 antibacterial therapeutics. Per its charter, the fund prioritizes investment in treatments that address a pathogen prioritized by the WHO, the CDC and/or other public health entities that: (i) are novel (e.g., absence of known cross-resistance, novel targets, new chemical classes, or new mechanisms of action); and/or (ii) have significant differentiated clinical utility (e.g., differentiated innovation that provides clinical value versus standard of care to prescribers and patients, such as safety/tolerability, oral formulation, different spectrum of activity); and (iii) reduce patient mortality. It is also expected that such agents would have the potential to strongly address the likely requirements for delinked Pull incentives such as the UK (NHS England) subscription pilot and the PASTEUR Act in the US. Submit queries to contact@amractionfund.com.
- INCATE (Incubator for Antibacterial Therapies in Europe) is an early-stage funding vehicle supporting innovation vs. drug-resistant bacterial infections. The fund provides advice, community, and non-dilutive funding (€10k in Stage I and up to €250k in Stage II) to support early-stage ventures in creating the evidence and building the team needed to get next-level funding. Details and contacts on their website (https://www.incate.net/).
- These things aren’t sources of funds but would help you develop funding applications
- The Global AMR R&D Hub’s dynamic dashboard (link) summarizes the global clinical development pipeline, incentives for AMR R&D, and investors/investments in AMR R&D. See also the 7 Feb 2026 newsletter (“The global funding pipeline, 2017-2023: A review”) about an excellent deep dive by the Hub team into patterns of funding over time.
- Antimicrobial Resistance Research and Innovation in Australia is an actively updated summary that covers Australia’s AMR research and patent landscape. It is provided via collaboration between The Lens (an ambitious project seeking to discover, analyse, and map global innovation knowledge) and CSIRO (Commonwealth Scientific and Industrial Research Organisation, an Australian Government agency responsible for scientific research). Lots to explore here!
- Diagnostic developers would find valuable guidance in this 6-part series on in vitro diagnostic (IVD) development. Sponsored by CARB-X, C-CAMP, and FIND, it pulls together real-life insights into a succinct set of tutorials.
- In addition to the lists provided by the Global AMR R&D Hub, you might also be interested in my most current lists of R&D incentives (link) and priority pathogens (link).